FDA approves two more COVID-19 vaccines for use in Ghana
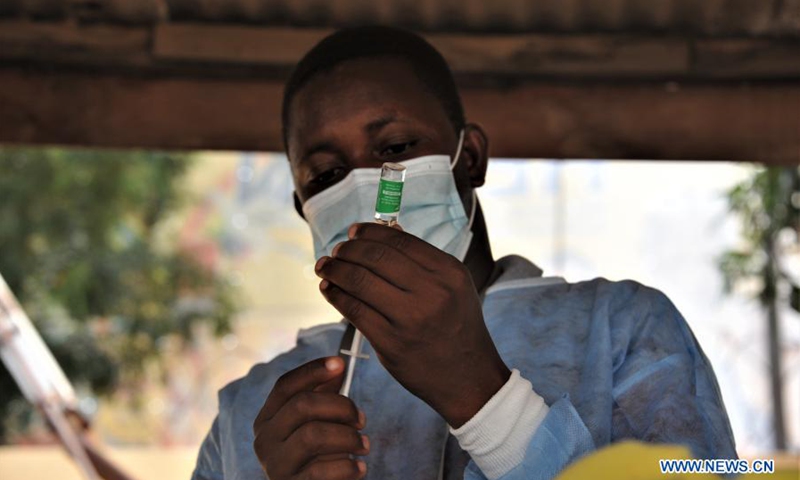
The Food and Drugs Authority (FDA) has approved two (2) more COVID-19 vaccines for importation, distribution, and use in Ghana.
The two new vaccines are Moderna (Spikevax) and Pfizer-BioNTech (Comirnaty) bringing the total number of approved COVID-19 vaccines for use in Ghana to five.
The FDA in a statement said it continues to use the Emergency Use Authorization (EUA) pathway, which is the accepted procedure for all national regulatory authorities globally, to ensure timely access to quality and safe medical products including vaccines during this pandemic.
The Authority assured the public that all the approved COVID-19 vaccines have been subjected to rigorous safety parameters and urged them to receive the vaccines without any reservations.
“The FDA remains committed to safeguarding the health and safety of the public as mandated by the Public Health Act, 2012 (Act 851),” the statement added.
COVID-19 vaccination programme to resume soon – Akufo-Addo
President Nana Addo Dankwa Akufo-Addo recently said Ghana will soon resume its vaccination programme.
Calls for the government to resume the vaccination have increased, following a spike in COVID-19 cases.
Speaking at a meeting with the outgoing Spanish Ambassador to Ghana on Sunday, July 25, 2021, President Akufo-Addo said Ghana had procured some vaccines that will facilitate the vaccination programme.
“The shortage of vaccines has been a problem for countries like ours, but hopefully we will see light at the end of the tunnel very soon. We will start the process of vaccinating or revaccinating our population.”
Ghana is expecting to take stock of over 17 million doses of vaccines from different sources within the third quarter.
Click here to read the full statement from the FDA
Source: Citinewsroom








